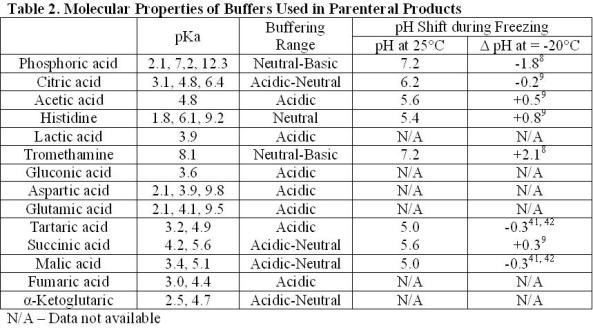
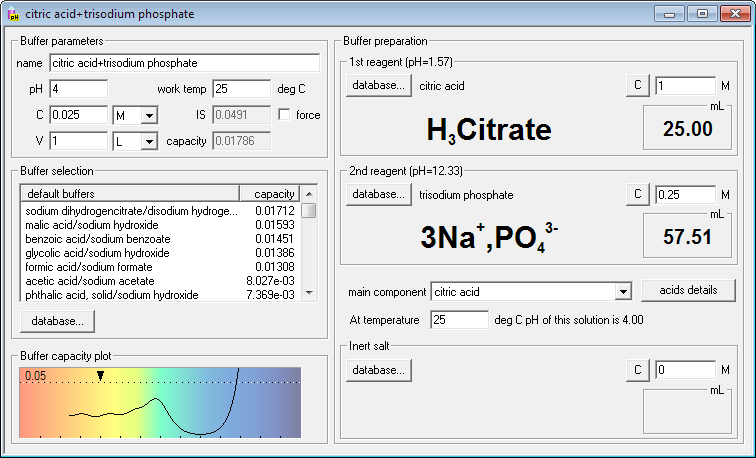
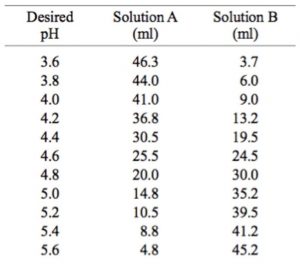
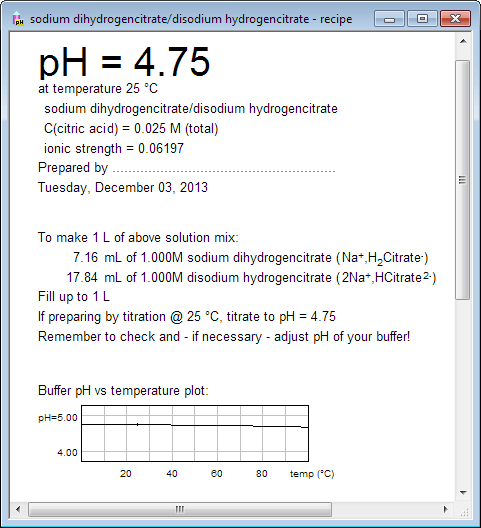
Rob Beynon 🏴FLSW on Twitter: "Want to create recipes for thermodynamically correct buffers? Try my buffer calculator here! My most impactful output - help me get to 1million litres before I retire!

SOLVED: Calculate the amount of monobasic potassium phosphate needed to prepare 100 mL of a 1 M solution. Molecular formula: KH2PO4 Molecular Mass: 136.09 g/mol Calculate the amount of dibasic potassium phosphate
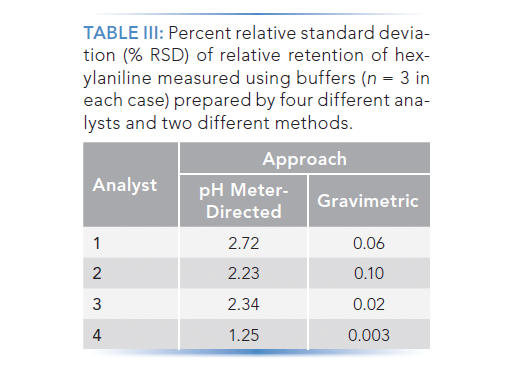
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability
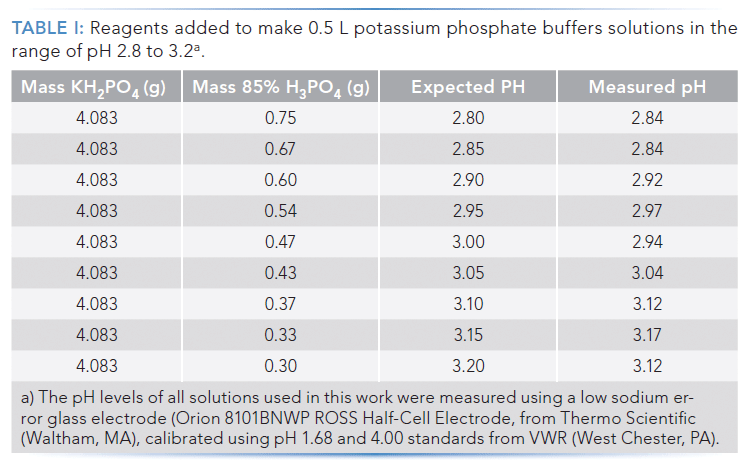
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability

SOLVED: Calculate the amount of monobasic potassium phosphate needed to prepare 100 mL of a 1 M solution. Molecular formula: KH2PO4 Molecular Mass: 136.09 g/mol Calculate the amount of dibasic potassium phosphate